Opportunity
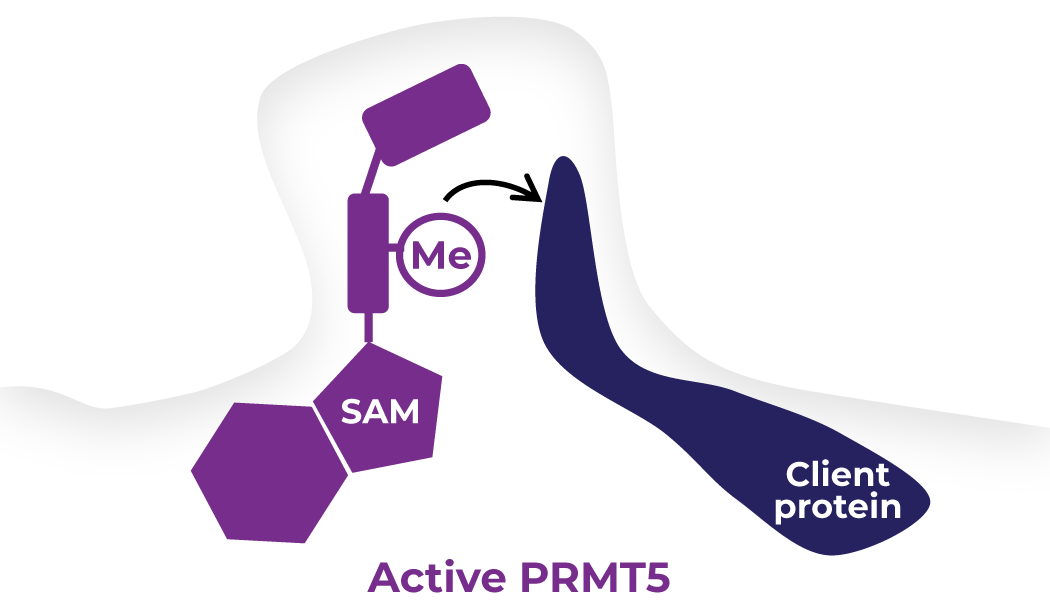
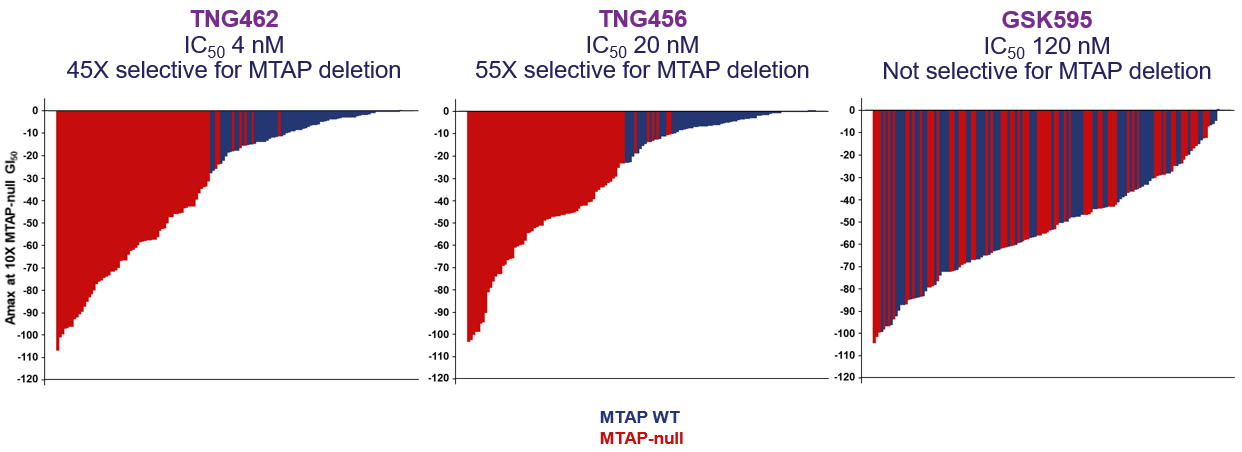
Vopimetostat and TNG456 are potent synthetic lethal inhibitors of PRMT5 (protein arginine methyl transferase 5) that work selectively in cancer cells with MTAP (methylthioadenosine phosphorylase) deletion, a common genetic alteration in a number of cancer types that does not occur in normal cells. Loss of MTAP leads to an accumulation of the inhibitory PRMT5 co-factor MTA (5’-deoxy-5’-methylthioadenosine). This increase in MTA markedly reduces PRMT5 activity in MTAP-deleted cancer cells but not in normal cells, and results in a vulnerability that makes MTAP-deleted cells more susceptible to PRMT5 inhibition than normal cells.
MTAP deletions occur in 10-15% of all human cancers, including many common cancers with limited treatment options such as non-small cell lung cancer (both squamous and non-squamous), mesothelioma, pancreatic cancer, cholangiocarcinoma, and glioblastoma (GBM) creating a potentially very significant therapeutic opportunity for patients.
In addition, MTAP deletions are frequently co-deleted with CDKN2A, which may sensitize CDKN2A-deleted tumors to CDK4/6 inhibition. Based on strong preclinical data showing significant in vivo combination benefit with CDKN2A deletions and other co-occurring genetic alterations, such as KRAS mutations and EGFR mutations, we are evaluating strategies for potential clinical combinations of our PRMT5 inhibitors with other oncogene-targeted therapies.