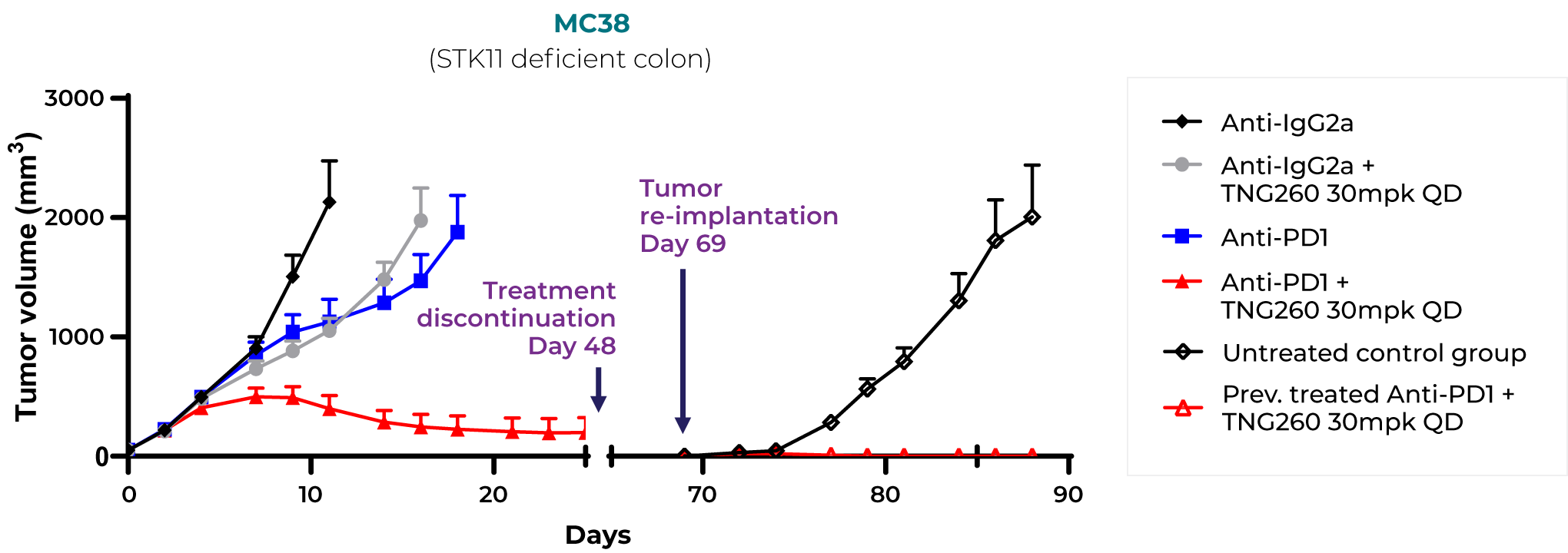
The remarkable clinical activity of immuno-oncology therapies, specifically checkpoint inhibitors, comes from activating certain immune cells to kill cancer cells. However, loss-of-function mutations in STK11 and some other tumor suppressor genes are strongly associated with immune checkpoint inhibitor resistance. TNG260 is a novel small molecule inhibitor of the Co-repressor of Repressor Element-1 Silencing Transcription (CoREST) deacetylase complex. In combination with immune checkpoint blockade, this CoREST inhibitor is designed to address the unmet medical need of immunotherapy-resistant STK11-mutant cancers.
OPPORTUNITY
Our synthetic lethal target discovery approach can be adapted to identify druggable tumor-intrinsic targets that do not kill cancer cells directly, but rather attract immune cells to destroy them. In preclinical studies we have shown that CoREST inhibition reverses the immune evasion effect of STK11 (serine-threonine kinase 11) loss-of-function mutations, which are present in approximately 15% of non-squamous lung and cervical cancers, 10% of carcinoma of unknown primary, 5% of breast cancers and 3% of pancreatic cancers.