A critical need
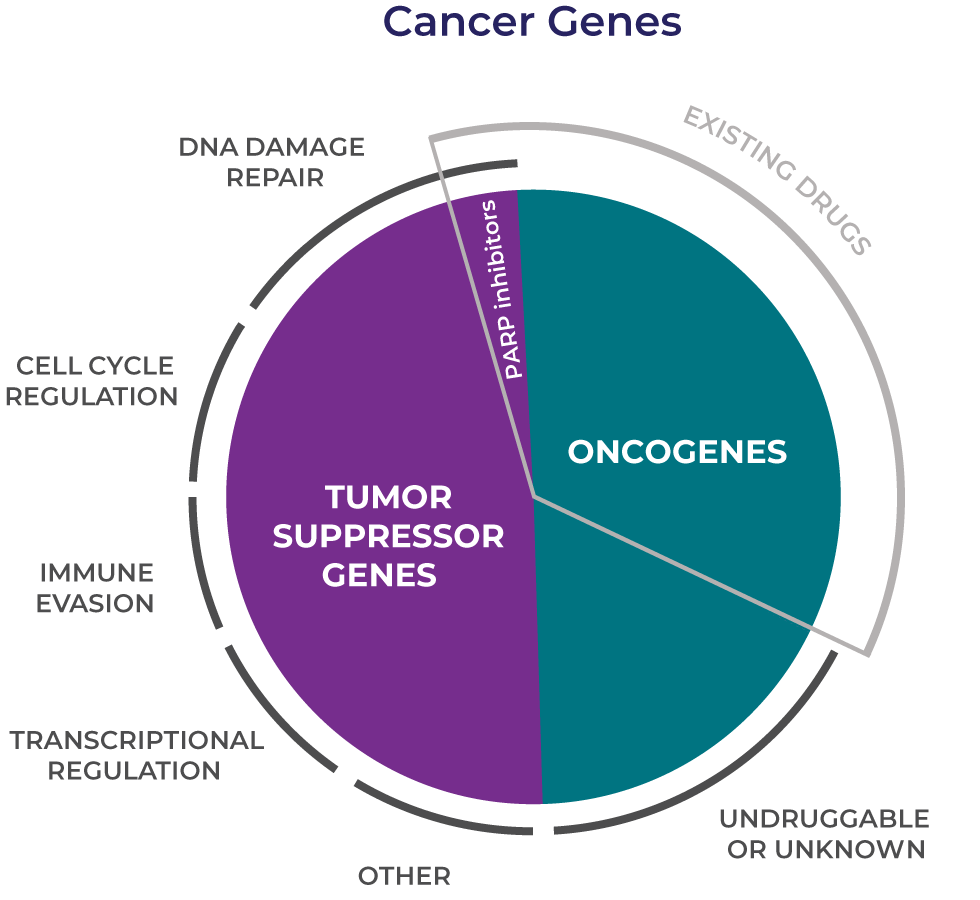
Great strides have been made in understanding the genetic drivers of cancer, but too many patients still lack effective treatment. Existing therapies target many of the activated oncogenes responsible for tumor growth but do not address the genetic alterations that fuel cancer by inactivating tumor suppressor genes and helping cancer cells evade killing by the immune system.
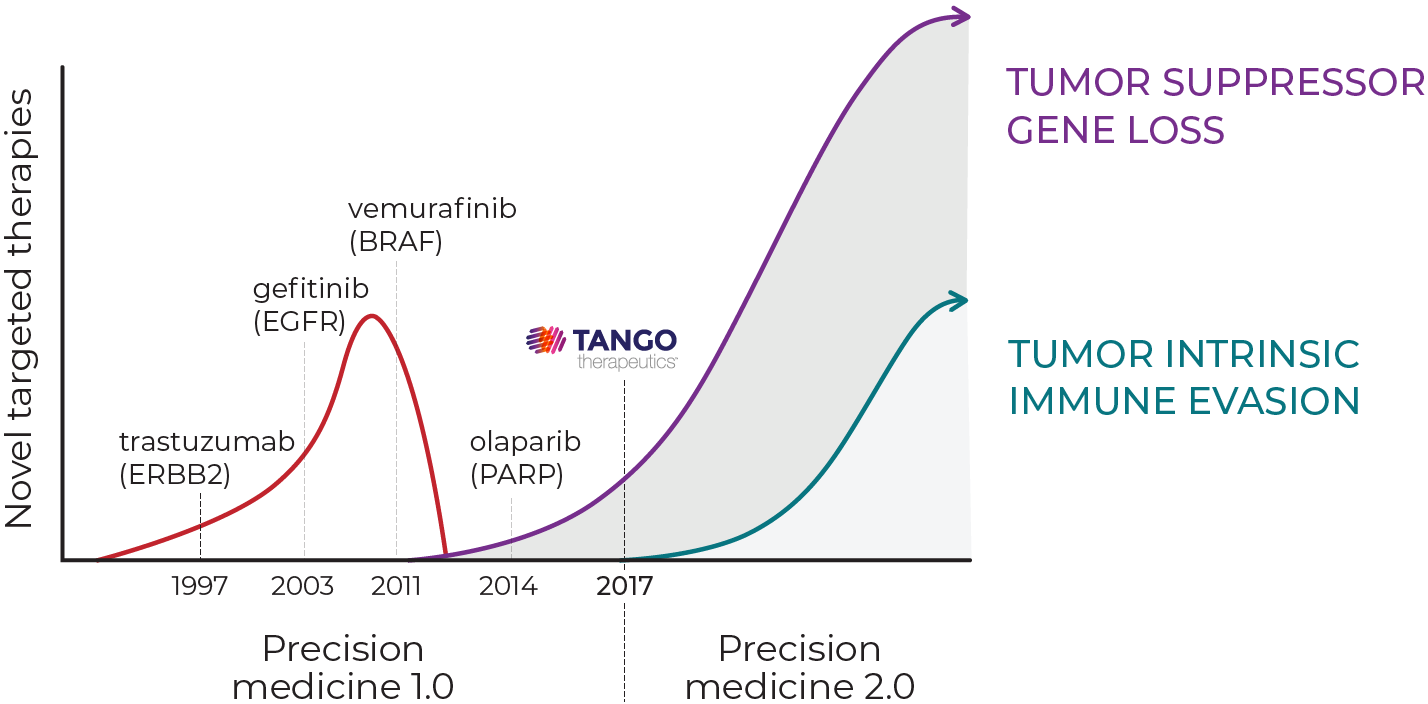
The next wave of targeted therapies
The first wave of targeted therapies revolutionized cancer care for patients whose cancers have specific activating mutations. These drugs target oncogenes such as BRAF, EGFR and ERBB2. However, genetic alterations that inactivate tumor suppressor genes are just as important in initiating and maintaining cancer growth and cannot be directly targeted.
Tango was founded in 2017 as a precision medicine company to address this broad unmet need and deliver the next wave of targeted therapies. We built a state-of-the-art CRISPR-based functional genomics target discovery platform and a powerful bio-analytic pipeline that integrate targets, pathways, genetic context and functional data to identify novel synthetic lethal targets for loss or inactivation of multiple tumor suppressor genes.